Why Are Lipid Nanoparticles So Important to the COVID-19 Vaccine?
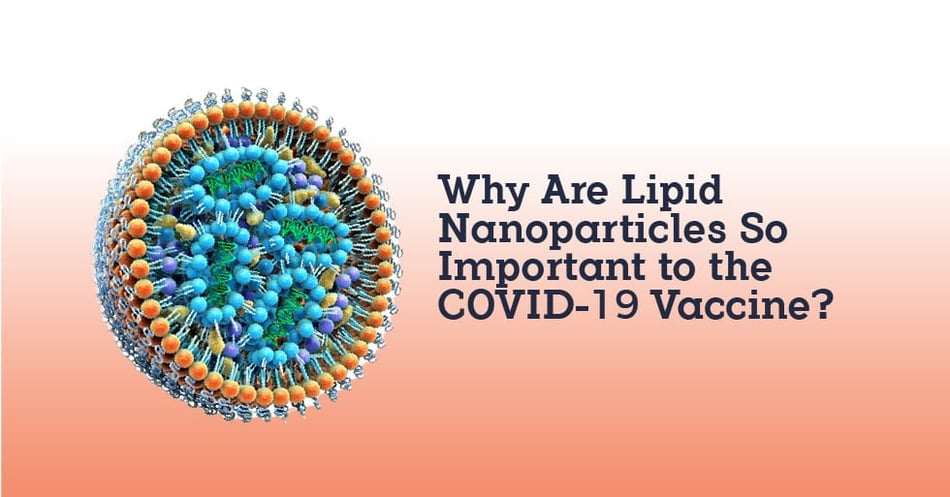
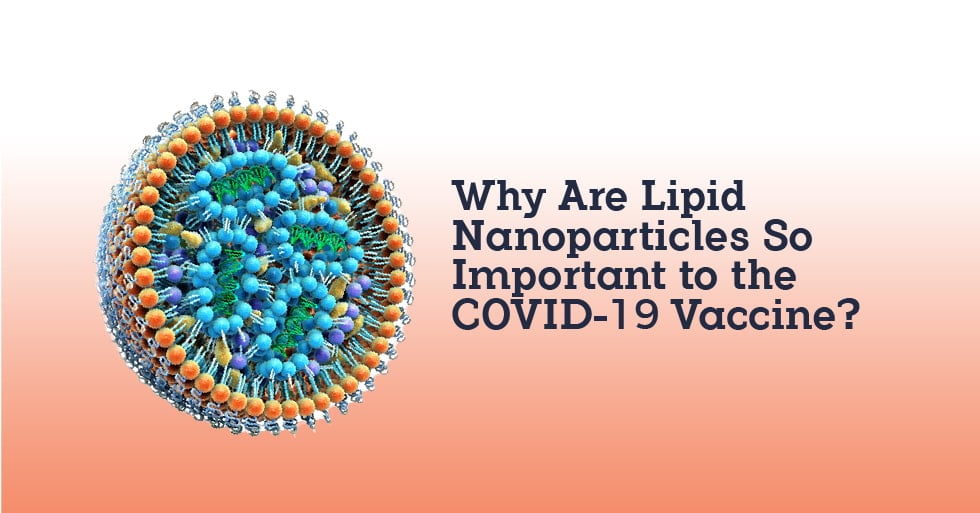 A recent federal report and subsequent media coverage has revealed an urgent gap in the supply chain for the Pfizer-BioNTech vaccine: lipid nanoparticles. Learning about this critical component is an excellent way to learn more about how the mRNA vaccine created by Pfizer and Moderna work. In today's post, we'll look at this important ingredient and steps being taken to make sure production is ramped up.
A recent federal report and subsequent media coverage has revealed an urgent gap in the supply chain for the Pfizer-BioNTech vaccine: lipid nanoparticles. Learning about this critical component is an excellent way to learn more about how the mRNA vaccine created by Pfizer and Moderna work. In today's post, we'll look at this important ingredient and steps being taken to make sure production is ramped up.
Let's start with a quick overview of the types of vaccines we have used for decades. Attenuated vaccines (measles) put a weak virus into our bodies, allowing our immune system to create antibodies without risk of a severe infection. These types of vaccines take a long time to create, since getting them into their weaker form requires that they be processed multiple times. Deactivated vaccines (polio) also take a longer time to produce in order to deactivate and prepare the virus for injection. Toxoid (diphtheria) vaccines and subunit vaccines (Hepatitis B) also take a long time to develop, as they each isolate parts of the virus and then require a vector (usually a harmless virus of another species) to enter the body and trigger the required immune response.
The COVID-19 vaccines currently in global distribution are known as mRNA vaccines. These vaccines deliver mRNA to our cells, causing the cells to essentially produce the vaccine inside our bodies. Unlike all the others, mRNA vaccines can be produced quickly: Once the virus is sequenced (getting the DNA code), the portion of it that creates the spike protein is identified and then quickly reproduced using polymerase chain reaction technology. The challenge in mRNA vaccines, however, is packaging that RNA, the "instruction" version of the DNA, so that our cells accept it. First, mRNA is supposed to stay INSIDE the cell, so our cell walls are constructed to keep mRNA from passing into or out of the cell. One of the ways this is prevented is by carrying an electrical charge that will repel invaders (polarity). Second, and in response to the first, our bodies have enzymes whose sole job is to destroy wandering mRNA. This is how our bodies protect us from getting some kind of foreign plant or animal mRNA into our cells.
So how do we trick the body into allowing the mRNA vaccine to travel safely through our bodies and then enter our cells? We package it in a lipid nanoparticle shell, a fatty biological material that will not attract the destructive enzymes, and most spectacularly, that has the charge required to be allowed into the cell and then release the mRNA into the cell. Manufacturing this type of lipid, an ionizable cationic lipid layer, is currently not keeping up with demand.
Without this lipid layer, the mRNA vaccine is not possible. The issue with the vaccine supply chain is that only one independent company makes this lipid layer, supplying Pfizer with its raw material. (Moderna has its own lipid production capabilities, and is also trying to scale quickly to meet demand.) The Biden administration has identified this specific supply chain issue as an "urgent gap" requiring immediate support from federal agencies, including the use of the Defense Production Act. This kind of support is intended to help the scaling up of production for this specific material to prevent vaccine bottlenecks.
The lipid nanoparticles are not the only urgent gaps receiving support. Besides the expected items like N95 masks and other protective gear, supplies such as swabs, pipettes, reagents, and a special type of syringe needle that can extract extra doses from vials, are also included. As the nation mobilizes to get individuals to get their vaccine, the efforts behind the scenes to make sure there are enough vaccines is astronomical. And the demand is huge: currently, 72.5 million doses have been delivered by Pfizer and Moderna combined, but Pfizer alone expects to produce 2 billion doses by the end of the year. And that's a lot of ionizable cationic lipid nanoparticles.
![EOScu Logo - Dark - Outlined [07182023]-01](https://blog.eoscu.com/hubfs/Eoscu_June2024/Images/EOScu%20Logo%20-%20Dark%20-%20Outlined%20%5B07182023%5D-01.svg)




